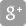B. Braun launches the First FDA-Approved Heparin Sodium Prefilled Syringe with attached safety needle
BETHLEHEM, Pa., April 24, 2019 (GLOBE NEWSWIRE) -- B. Braun Medical Inc. announced today the launch of its Heparin Sodium Injection, USP - the first Heparin 5,000 USP Unit / 0.5 mL prefilled syringe with attached safety needle for subcutaneous and intravenous use in the United States.
“B. Braun continues to develop products designed to increase patient and clinician safety, while reducing medication errors and improving dosage accuracy and workflows,” said Leigh Nickens, Director of Marketing, Fluid Therapy and Injectable Drugs at B. Braun. “Our prefilled syringe is equipped with a label-integrated Schreiner MediPharm Needle-Trap system that, upon activation, guards against accidental needle stick injuries,” Nickens continued.
The Heparin Sodium Injection, USP prefilled syringe is preservative free, not made with natural rubber latex, and has 24-month stability at room temperature.
Heparin Sodium Injection, USP is indicated for: prophylaxis and treatment of venous thrombosis and pulmonary embolism, prevention of postoperative deep venous thrombosis and pulmonary embolism in patients undergoing major abdominothoracic surgery or who, for other reasons, are at risk of developing thromboembolic disease, atrial fibrillation with embolization, treatment of acute and chronic consumptive coagulopathies (disseminated intravascular coagulation), prevention of clotting in arterial and cardiac surgery, prophylaxis and treatment of peripheral arterial embolism, anticoagulant use in blood transfusions, extracorporeal circulation, and dialysis procedures.
For additional information, please see full Prescribing Information.
IMPORTANT SAFETY INFORMATION
Contraindications:
- Severe thrombocytopenia
- When suitable blood coagulation tests, e.g., the whole blood clotting time, partial thromboplastin time, etc., cannot be performed at appropriate intervals
- An uncontrolled active bleeding state, except when this is due to disseminated intravascular coagulation
About B. Braun
B. Braun Medical Inc., a leader in infusion therapy and pain management, develops, manufactures, and markets innovative medical products and services to the healthcare industry. Other key product areas include nutrition, pharmacy admixture and compounding, ostomy and wound care, and dialysis. The company is committed to eliminating preventable treatment errors and enhancing patient, clinician and environmental safety. B. Braun Medical is headquartered in Bethlehem, Pa., and is part of the B. Braun Group of Companies in the U.S., which includes B. Braun Interventional Systems, Aesculap® and CAPS®.
Globally, the B. Braun Group of Companies employs more than 61,000 employees in 64 countries. Guided by its Sharing Expertise® philosophy, B. Braun continuously exchanges knowledge with customers, partners and clinicians to address the critical issues of improving care and lowering costs. To learn more about B. Braun Medical, visit www.BBraunUSA.com.
About Schreiner MediPharm
Based in the Munich area of Germany, Schreiner MediPharm is a global leader in the development and manufacture of innovative specialty labels with value-added benefits that have set the standard in the healthcare and pharmaceutical industries. Schreiner MediPharm’s expertise is based on over 65 years of experience. The family-owned company provides specialty labels and label printing services to the biggest names in the healthcare market in over 20 countries around the world, including Japan.
Schreiner MediPharm’s U.S. facility, a business unit of Schreiner Group LP, is located in Blauvelt, NY.
| Contact: | Allison Longenhagen B. Braun Medical Inc. 610.997.4768 allison.longenhagen@bbraunusa.com | |

Upcoming Life Sciences Events
- April 2024
- London: BioTrinity 2024
- Singapore: Asia Bio Partnering Forum
- London: LSX World Congress
 Biotechgate Global Database ›
Biotechgate Global Database ›